|
Glossary Term - 1. Most Abundant Compounds in the Earth's Crust. Most Abundant Compounds in the Earth's Crust. Source: Exploring Chemical Elements and their Compounds; David L.
Heiserman, 1. 99. Compound. Formula. Abundancepercent by weight. Abundanceparts per million by weight. Silicon dioxide. Si. O2. 42. 8. 6%4. 28,6. A worksheet on writing formulas for ionic compounds. A fun and exciting activity for naming chemical compounds. Naming compounds is one of the hardest. This tutorial introduces chemical compounds. Other sections include matter, elements, the periodic table, reactions, and biochemistry. Magnesium oxide. Mg. O3. 5. 0. 7%3. 50,7. Ferrous oxide. Fe. O8. 9. 7%8. 9,7. 00. Aluminum oxide. Al. O3. 6. 9. 9%6. 9,9. Calcium oxide. Ca. O4. 3. 7%4. 3,7. 00. Sodium oxide. Na. Example 3 Write the chemical formula for an ionic compound composed of each pair of ions. Sodium is a chemical element with symbol Na (from Latin natrium) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal. Sodium hydroxide (NaOH), also known as lye and caustic soda, is an inorganic compound. It is a white solid and highly caustic metallic base and alkali of sodium which. Search the database of common compounds in chemistry and get the molecular weight of any chemical formula. Teacher resources and professional development across the curriculum. Teacher professional development and classroom resources across the curriculum. O0. 4. 5%4,5. 00. Ferric oxide. Fe. O3. 0. 3. 6%3,6. 00. 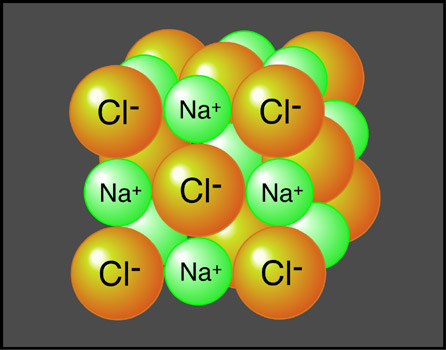
Titanium dioxide. Ti. O2. 0. 3. 3%3,3. Chromic oxide. Cr. O3. 0. 1. 8%1,8. 00. Manganese dioxide. Mn. O2. 0. 1. 4%1,4. Ways to Name Chemical Compounds. There are two types of acids: those that contain oxygen, and those that do not. 
Sodium hydroxide - Wikipedia. Sodium hydroxide. Names. Preferred IUPAC name. Systematic IUPAC name. Other names. Caustic soda. Lye. It is a white solid and highly caustic metallic base and alkali of sodium which is available in pellets, flakes, granules, and as prepared solutions at different concentrations. This alkali is deliquescent and readily absorbs moisture and carbon dioxide in air. Sodium hydroxide is used in many industries, mostly as a strong chemicalbase in the manufacture of pulp and paper, textiles, drinking water, soaps and detergents and as a drain cleaner. Worldwide production in 2. It is highly soluble in water, with a lower solubility in ethanol and methanol, but is insoluble in ether and other non- polar solvents. Similar to the hydration of sulfuric acid, dissolution of solid sodium hydroxide in water is a highly exothermic reaction. The resulting solution is usually colourless and odorless. As with other alkaline solutions, it feels slippery when it comes in contact with skin. Chemical properties. For example, when sodium hydroxide reacts with hydrochloric acid, sodium chloride is formed: Na. OH(aq) + HCl(aq) . Such acid- base reactions can also be used for titrations. However, sodium hydroxide is not used as a primary standard because it is hygroscopic and absorbs carbon dioxide from air. Reaction with acidic oxides. Such reactions are often used to . For example,2 Na. OH + SO2 . Because of this, glass joints and stopcocks exposed to sodium hydroxide have a tendency to . Flasks and glass- lined chemical reactors are damaged by long exposure to hot sodium hydroxide, which also frosts the glass. Sodium hydroxide does not attack iron since iron does not have amphoteric properties (i. A few transition metals, however, may react vigorously with sodium hydroxide. In 1. 98. 6, an aluminium road tanker in the UK was mistakenly used to transport 2. The pressurization was due to the hydrogen gas which is produced in the reaction between sodium hydroxide and aluminium: 2 Al + 2 Na. OH + 6 H2. O . The following colours are observed: blue- copper, green- iron(II), yellow/brown- iron(III). Zinc and lead salts dissolve in excess sodium hydroxide to give a clear solution of Na. Zn. O2 or Na. 2Pb. O2. Aluminium hydroxide is used as a gelatinous flocculant to filter out particulate matter in water treatment. Aluminium hydroxide is prepared at the treatment plant from aluminium sulfate by reacting it with sodium hydroxide or bicarbonate. Al. 2(SO4)3 + 6 Na. OH . However, the limited solubility of sodium hydroxide in organic solvents means that the more solublepotassium hydroxide (KOH) is often preferred. Touching sodium hydroxide solution with the bare hands, while not recommended, produces a slippery feeling. This happens because the oils of the hand are converted to soap. Production. Chlorine gas is also produced in this process. Solid sodium hydroxide is obtained from this solution by the evaporation of water. Solid sodium hydroxide is most commonly sold as flakes, prills, and cast blocks. North America and Asia each contributed around 1. Europe produced around 1. In the United States, the major producer of sodium hydroxide is the Dow Chemical Company, which has annual production around 3. Freeport, Texas, and Plaquemine, Louisiana. Other major US producers include Oxychem, PPG, Olin, Pioneer Companies, Inc. All of these companies use the chloralkali process. The byproducts are hydrogen gas and heat, often resulting in a flame, making this a common demonstration of the reactivity of alkali metals in academic environments; however, it is not commercially viable, as the isolation of sodium metal is typically performed by reduction or electrolysis of sodium compounds including sodium hydroxide. For further information in historical production, see alkali manufacture. Around 5. 6% of sodium hydroxide produced is used by industry, 2. Sodium hydroxide is also used in the manufacture of sodium salts and detergents, p. H regulation, and organic synthesis. It is used in the Bayer process of aluminium production. As above, sodium hydroxide reacts with weak acids such as hydrogen sulfide and mercaptans to yield non- volatile sodium salts, which can be removed. The waste which is formed is toxic and difficult to deal with, and the process is banned in many countries because of this. In 2. 00. 6, Trafigura used the process and then dumped the waste in Africa. Along with sodium sulfide, sodium hydroxide is a key component of the white liquor solution used to separate lignin from cellulosefibers in the kraft process. It also plays a key role in several later stages of the process of bleaching the brown pulp resulting from the pulping process. These stages include oxygen delignification, oxidative extraction, and simple extraction, all of which require a strong alkaline environment with a p. H > 1. 0. 5 at the end of the stages. Tissue digestion. This process involved placing a carcass into a sealed chamber, then adding a mixture of sodium hydroxide and water (which breaks the chemical bonds that keep the flesh intact). This eventually turns the body into a liquid with coffee- like appearance. Italian serial killer. Leonarda Cianciulli used this chemical to turn dead bodies into soap. If a dilute solution is spilled on the skin, burns may result if the area is not washed thoroughly and for several minutes with running water. Splashes in the eye can be more serious and can lead to blindness. Sodium hydroxide reacts with aluminium and water to release hydrogen gas. The aluminium takes the oxygen atom from sodium hydroxide, which in turn takes the oxygen atom from the water, and releases the two hydrogen atoms, The reaction thus produces hydrogen gas and sodium aluminate. In this reaction, sodium hydroxide acts as an agent to make the solution alkaline, which aluminium can dissolve in. This reaction can be useful in etching, removing anodizing, or converting a polished surface to a satin- like finish, but without further passivation such as anodizing or alodining the surface may become degraded, either under normal use or in severe atmospheric conditions. In the Bayer process, sodium hydroxide is used in the refining of alumina containing ores (bauxite) to produce alumina (aluminium oxide) which is the raw material used to produce aluminium metal via the electrolytic. Hall- H. Since the alumina is amphoteric, it dissolves in the sodium hydroxide, leaving impurities less soluble at high p. H such as iron oxides behind in the form of a highly alkaline red mud. Other amphoteric metals are zinc and lead which dissolve in concentrated sodium hydroxide solutions to give sodium zincate and sodium plumbate respectively. Esterification and transesterification reagent. This only works with anhydrous sodium hydroxide, because combined with water the fat would turn into soap, which would be tainted with methanol. Na. OH is used more often than potassium hydroxide because it is cheaper and a smaller quantity is needed. Food preparation. Owing to the difficulty in obtaining food grade sodium hydroxide in small quantities for home use, sodium carbonate is often used in place of sodium hydroxide. These expand considerably in size and may be further processed by frying to make corn nuts or by drying and grinding to make grits. Nixtamal is similar, but uses calcium hydroxide instead of sodium hydroxide. Sodium hydroxide is also the chemical that causes gelling of egg whites in the production of Century eggs. German pretzels are poached in a boiling sodium carbonate solution or cold sodium hydroxide solution before baking, which contributes to their unique crust. Lye- water is an essential ingredient in the crust of the traditional baked Chinese moon cakes. Most yellow coloured Chinese noodles are made with lye- water but are commonly mistaken for containing egg. Some methods of preparing olives involve subjecting them to a lye- based brine. A similar process is also used in the kakanin known as pitsi- pitsi or pichi- pichi except that the mixture uses grated cassava instead of rice flour. Cleaning agent. It is added to water, heated, and then used to clean process equipment, storage tanks, etc. It can dissolve grease, oils, fats and protein- based deposits. It is also used for cleaning waste discharge pipes under sinks and drains in domestic properties. Surfactants can be added to the sodium hydroxide solution in order to stabilize dissolved substances and thus prevent redeposition. A sodium hydroxide soak solution is used as a powerful degreaser on stainless steel and glass bakeware. It is also a common ingredient in oven cleaners. A common use of sodium hydroxide is in the production of parts washerdetergents. Parts washer detergents based on sodium hydroxide are some of the most aggressive parts washer cleaning chemicals. The sodium hydroxide- based detergents include surfactants, rust inhibitors and defoamers. A parts washer heats water and the detergent in a closed cabinet and then sprays the heated sodium hydroxide and hot water at pressure against dirty parts for degreasing applications. Sodium hydroxide used in this manner replaced many solvent- based systems in the early 1. Water and sodium hydroxide detergent- based parts washers are considered to be an environmental improvement over the solvent- based cleaning methods. The alkali dissolves greases to produce water solubleproducts. It also hydrolyzes the proteins such as those found in hair which may block water pipes. These reactions are sped by the heat generated when sodium hydroxide and the other chemical components of the cleaner dissolve in water. Such alkaline drain cleaners and their acidic versions are highly corrosive and should be handled with great caution. Sodium hydroxide is used in some relaxers to straighten hair. However, because of the high incidence and intensity of chemical burns, manufacturers of chemical relaxers use other alkaline chemicals in preparations available to average consumers. Chem. 4Kids. com: Atoms: Compounds. Compound Basics. Let’s start with molecules. Molecule is the general term used to describe any atoms that are connected by chemical bonds. Every combination of atoms is a molecule. A compound is a molecule made of atoms from different elements. All compounds are molecules, but not all molecules are compounds. Hydrogen gas (H2) is a molecule, but not a compound because it is made of only one element. Water (H2. O) can be called a molecule or a compound because it is made of hydrogen (H) and oxygen (O) atoms. Atoms that share electrons in a chemical bond have covalent bonds. An oxygen molecule (O2) is a good example of a molecule with a covalent bond. Ionic bonds occur when electrons are donated from one atom to another. Table salt (Na. Cl) is a common example of a compound with an ionic bond. Metallic bonds occur between metal atoms. We’re going to focus on ionic and covalent bonds. Physical forces alone (unless you're inside of the Sun or something extreme) rarely break down compounds completely. You can apply heat to melt an ice cube, but there will be no change in the water molecules. You can also place a cup of water in a container and decrease the pressure. Forces act on the bonds between atoms, changing the molecular structure of a substance. You can pour liquid acid on a solid and watch the solid dissolve. That process is a chemical change because molecular bonds are being created and destroyed. Geologists pour acids on rocks to test for certain compounds. Probably everything you can see is one type of compound or another. When elements join and become compounds, they lose many of their individual traits. Sodium (Na) alone is very reactive. But when sodium and chlorine (Cl) combine, they form a non- reactive substance called sodium chloride (table salt, Na. Cl). New compounds have few or none of the physical or chemical traits of the original elements. They have a new life of their own. What about magnesium chloride (Mg. Cl. 2)? It contains one magnesium (Mg) and two chlorine (Cl) atoms. There are two ionic bonds. Methane (CH4) is made up of one carbon (C) and four hydrogen (H) atoms. There are four bonds and they are all covalent. However, most compounds have combinations of ionic and covalent bonds. Let's look at sodium hydroxide (Na- OH).. The bond that binds the hydrogen (H) to the oxygen (O) is covalent. The sodium is bonded to the hydroxide part of the compound with an ionic bond. This is a good example of how there can be different types of bonds within one compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
May 2017
Categories |
 RSS Feed
RSS Feed
